Human Powered AI
Remove the tedious, manual tasks that hold back your clinical staff. Say goodbye to bottlenecks that stop clinical studies in their tracks. BEKplatform is the AI-powered patient-matching solution that makes patient identification faster and more precise than ever before so that sites can confidently meet enrollment goals.
Why use the platform

10x more qualified patients
We speed up site feasibility and detect more protocol-eligible candidates leading to 10x more qualified patients and 2x more patients enrolled.

Data-driven study feasibility and recruitment commitments
We rapidly sift through structured and unstructured medical records, helping sites gain a clearer understanding of their patient population pre- and post-study award.

Save 100s of hours by eliminating unnecessary chart abstraction
We automate chart review to match more qualified potential participants, saving site coordinators hundreds of hours in the process.

How it works
Forever, the clinical trial enrollment process has relied on direct-to-consumer marketing or manual chart reviews, which is incredibly expensive, inefficient, and time consuming. Oftentimes resulting in a lot of guesswork and “hoping for the best”.
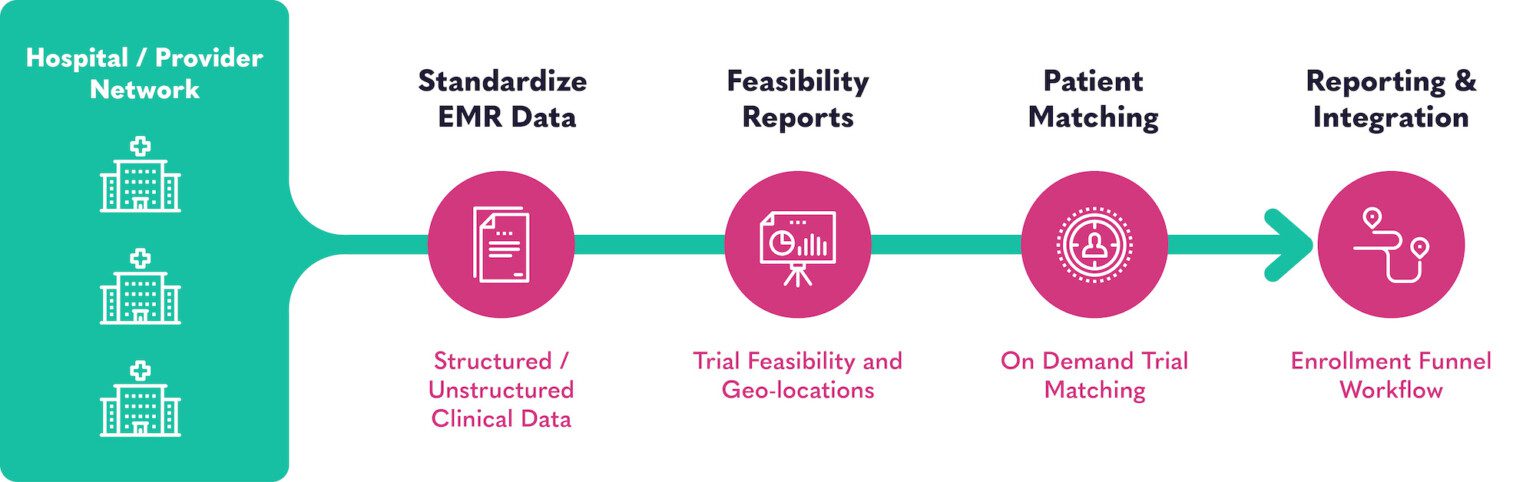
So, we built an AI-powered solution that instantaneously sorts and sifts through EMR data to find clinically-qualified patients for you to recruit. By translating structured and unstructured data (doctor notes, pdfs, lab reports, and clinical documents) the powerful AI translates EMR data from a system initially built for insurance to a purpose-built system for finding and matching patients to research studies.
Take a look inside BEKplatform, the AI-powered patient-matching technology for sites and healthcare organizations
Query and Cohort Builder
BEKplatform deciphers patient-level structured and unstructured medical data to detect more protocol-eligible candidates so you can build robust queries and patient cohorts. The platform ultimately turns disorganized, unstructured clinical data into a synthesized, longitudinal patient graph that is easily queryable.
Feasibility Reports and Insights
BEKplatform facilitates powerful decision making from easily digestible reports, allowing researchers to access real-time patient data and use it to analyze their patient populations, quickly determine trial feasibility, automate processes and identify untapped trial opportunities.
Turn unstructured data into searchable information
BEKplatform accesses the saved files, reports, and even hand-written notes associated with patient charts, then divides each note/record into sentences and transforms the underlying text.
The platform then analyzes and transforms text using deep learning neural net based on BERT to identify medical entities and associated attributes for key domains (ex: demographics, diagnosis, medications, etc.).
The text is then mapped to a searchable ontology made of more than 24 million search terms, synonyms, and lexemes. Combined with structured medical data, BEKplatfom generates a synthesized, longitudinal patient graph.
To support it's industry-leading 93% accuracy in interpreting EMR records, BEKhealth employs a human-in-the-loop feedback mechanism to hone the models’ outputs, ensuring a high level of reliability and relevance to clinical contexts.
Ingest
Automated ETL pipeline using 25+ proprietary EMR Adapters and 19+ NGS Processors

Trial Feasibility
AI-powered decision making, allowing researchers to access real-time patient data and use it to analyze their patient populations, quickly determine trial feasibility, automate processes and identify untapped trial opportunities (or avoid costly study startup miss-fires).
![]()
Unify
AI-powered data platform that standardizes structured and unstructured medical data by cleaning, interpreting, predicting, and validating your data.

Trial Candidates
AI-powered automated identification of patients that match I/E criteria with over 90% accuracy. Refreshed nightly, bringing new eligible patients and excluding ineligible patients from clinical research studies in real-time.

Smarter Recruitment. Faster Enrollment. Stronger Trials.
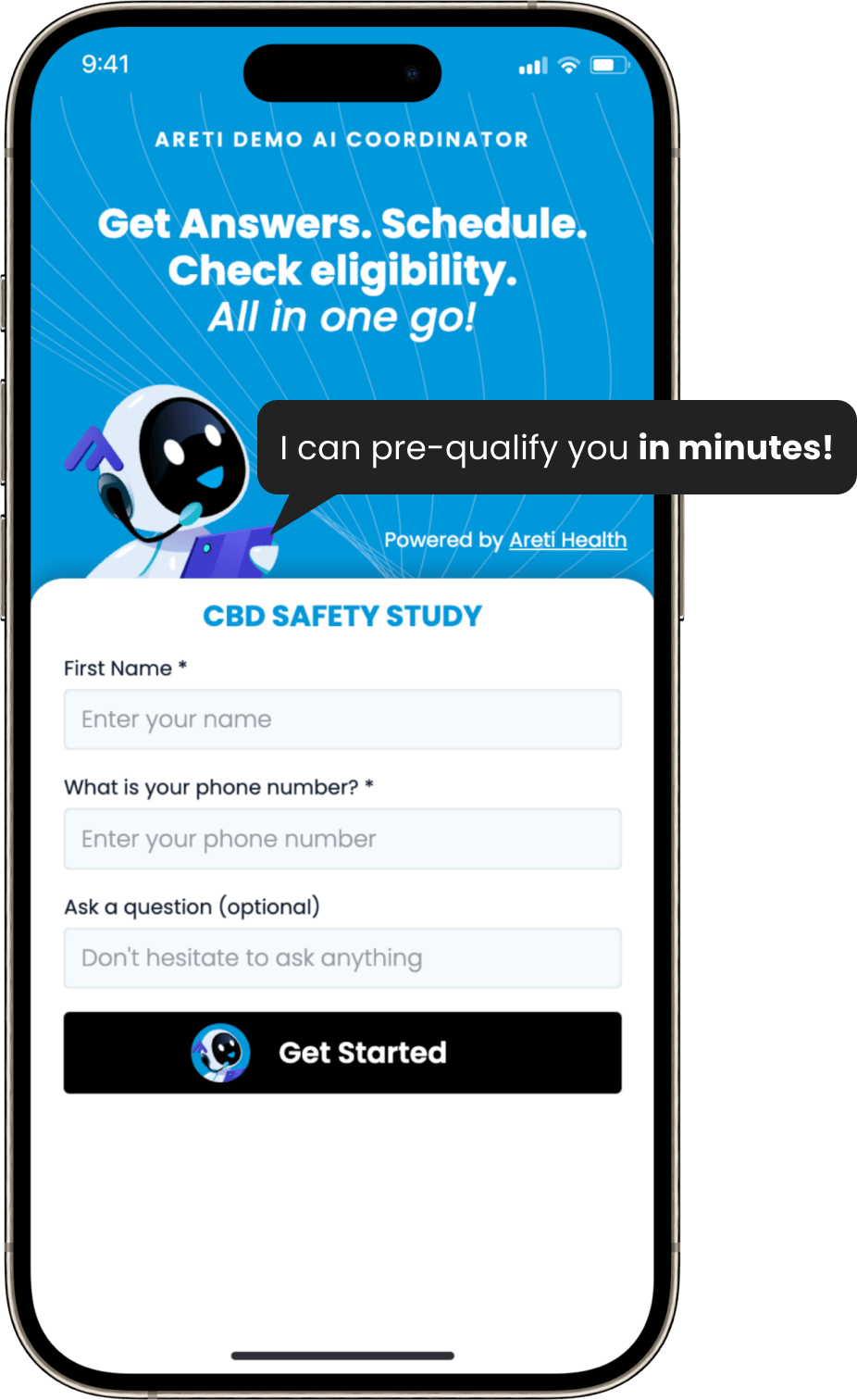
Areti Health and BEKhealth have teamed up to take the guesswork out of clinical trial recruitment and enrollment.
By combining BEKhealth’s AI-driven patient identification platform with Areti Health’s automated engagement and scheduling, we're delivering a seamless, end-to-end solution that accelerates enrollment, reduces site burden, and improves trial outcomes.
It’s a smarter, faster way to connect the right patients with the right studies—at exactly the right time.
Optimize
feasibility
Identify
eligible candidates
Accelerate
recruitment
60 Minutes After Implementation
our first joint client had...
200
clinically-eligible patients identified by BEKhealth
12
patients pre-screened by Areti Health
8
patients scheduled for appointments

Putting the human in AI
Don’t have the staff or expertise to build feasibility reports or trials? No problem, BEKhealth provides a specialized group of research nurses to assist your every move. We don’t just hand you a solution and send you on your way, we collaborate with hands-on guidance to ensure the best possible outcomes in your research.

Rapid Implementation
Our team works closely with you to implement and integrate the BEKplatform in an industry-leading 2-8 weeks. With 25+ existing EMR connectors, we can get you up and running faster to accelerate your research.
We currently support dozens of EMR platforms. Here are just a few on our ever growing list:

















Don’t see your EMR on the list?
No problem! We build new EMR connectors all the time. Our business development and clinical care teams can walk you through our process for ensuring your success.